In an period the place information is the lifeblood of medical development, the medical trial {industry} finds itself at a vital crossroads. The present panorama of medical information administration is fraught with challenges that threaten to stifle innovation and delay life-saving remedies.
As we grapple with an unprecedented deluge of knowledge—with a typical Part III trial now producing a staggering 3.6 million information factors, which is 3 times greater than 15 years in the past, and greater than 4000 new trials approved every year—our current information platforms are buckling beneath the pressure. These outdated techniques, characterised by information silos, poor integration, and overwhelming complexity, are failing researchers, sufferers, and the very progress of medical science. The urgency of this example is underscored by stark statistics: about 80% of medical trials face delays or untimely termination as a consequence of recruitment challenges, with 37% of analysis websites struggling to enroll sufficient contributors.
These inefficiencies come at a steep price, with potential losses starting from $600,000 to $8 million every day a product’s improvement and launch is delayed. The medical trials market, projected to succeed in $886.5 billion by 2032 [1], calls for a brand new technology of Medical Knowledge Repositories (CDR).
Reimagining Medical Knowledge Repositories (CDR)
Usually, medical trial information administration depends on specialised platforms. There are numerous causes for this, ranging from the standardized authorities’ submission course of, the consumer’s familiarity with particular platforms and programming languages, and the flexibility to depend on the platform vendor to ship area data for the {industry}.
With the worldwide harmonization of medical analysis and the introduction of regulatory-mandated digital submissions, it is important to know and function throughout the framework of world medical improvement. This entails making use of requirements to develop and execute architectures, insurance policies, practices, pointers, and procedures to handle the medical information lifecycle successfully.
A few of these processes embody:
- Knowledge Structure and Design: Knowledge modeling for medical information repositories or warehouses
- Knowledge Governance and Safety: Requirements, SOPs, and pointers administration along with entry management, archiving, privateness, and safety
- Knowledge High quality and Metadata administration: Question administration, information integrity and high quality assurance, information integration, exterior information switch, together with metadata discovery, publishing, and standardization
- Knowledge Warehousing, BI, and Database Administration: Instruments for information mining and ETL processes
These parts are essential for managing the complexities of medical information successfully.
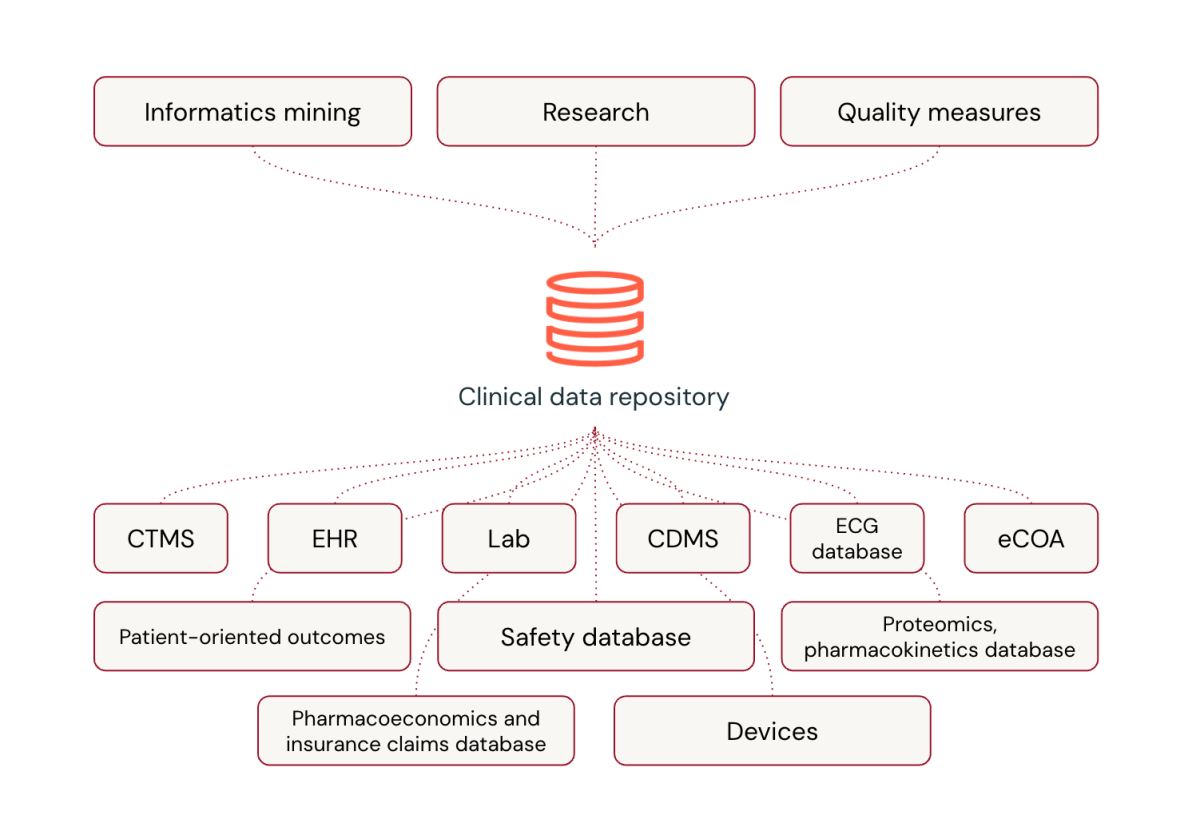
Common platforms are reworking medical information processing within the pharmaceutical {industry}. Whereas specialised software program has been the norm, common platforms supply important benefits, together with the flexibleness to include novel information sorts, close to real-time processing capabilities, integration of cutting-edge applied sciences like AI and machine studying, and strong information processing practices refined by dealing with large information volumes.
Regardless of issues about customization and the transition from acquainted distributors, common platforms can outperform specialised options in medical trial information administration. Databricks, for instance, is revolutionizing how Life Sciences firms deal with medical trial information by integrating numerous information sorts and offering a complete view of affected person well being.
In essence, common platforms like Databricks should not simply matching the capabilities of specialised platforms – they’re surpassing them, ushering in a brand new period of effectivity and innovation in medical trial information administration.
Leveraging the Databricks Knowledge Intelligence Platform as a basis for CDR
The Databricks Knowledge Intelligence Platform is constructed on prime of lakehouse structure. Lakehouse structure is a contemporary information structure that mixes the perfect options of information lakes and information warehouses. This corresponds nicely to the wants of the trendy CDR.
Though most medical trial information characterize structured tabular information, new information modalities like imaging and wearable units are gaining recognition. They’re the brand new method of redefining the medical trials course of. Databricks is hosted on cloud infrastructure, which supplies the flexibleness of utilizing cloud object storage to retailer medical information at scale. It permits storing all information sorts, controlling prices (older information may be moved to the colder tiers to avoid wasting prices however accommodate regulatory necessities of retaining information), and information availability and replication. On prime of this, utilizing Databricks because the underlying know-how for CDR permits one to maneuver to the agile improvement mannequin the place new options may be added in managed releases in opposition to Massive Bang software program model updates.
The Databricks Knowledge Intelligence Platform is a full-scale information platform that brings information processing, orchestration, and AI performance to at least one place. It comes with many default information ingestion capabilities, together with native connectors and probably implementing customized ones. It permits us to combine CDR with information sources and downstream purposes simply. This capacity gives flexibility and end-to-end information high quality and monitoring. Native assist of streaming permits to counterpoint CDR with IoMT information and acquire close to real-time insights as quickly as information is on the market. Platform observability is an enormous matter for CDR not solely due to strict regulatory necessities but in addition as a result of it allows secondary use of information and the flexibility to generate insights, which in the end can enhance the medical trial course of general. Processing medical information on Databricks permits for implementation of the versatile options to realize perception into the method. As an example, is processing MRI pictures extra resource-consuming than processing CT check outcomes?
Implementing a Medical Knowledge Repository: A Layered Strategy with Databricks
Medical Knowledge Repositories are subtle platforms that combine the storage and processing of medical information. Lakehouse medallion structure, a layered method to information processing, is especially well-suited for CDRs. This structure sometimes consists of three layers, every progressively refining information high quality:
- Bronze Layer: Uncooked information ingested from varied sources and protocols
- Silver Layer: Knowledge conformed to plain codecs (e.g., SDTM) and validated
- Gold Layer: Aggregated and filtered information prepared for assessment and statistical evaluation
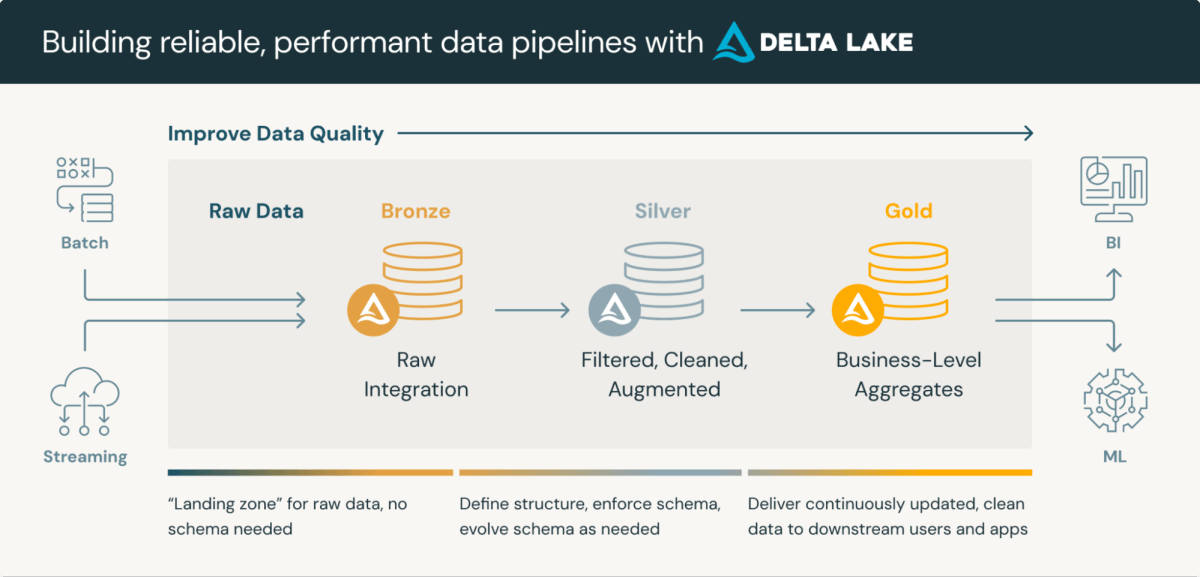
Using Delta Lake format for information storage in Databricks presents inherent advantages akin to schema validation and time journey capabilities. Whereas these options want enhancement to totally meet regulatory necessities, they supply a strong basis for compliance and streamlined processing.
The Databricks Knowledge Intelligence Platform comes geared up with strong governance instruments. Unity Catalog, a key part, presents complete information governance, auditing, and entry management throughout the platform. Within the context of CDRs, Unity Catalog allows:
- Monitoring of desk and column lineage
- Storing information historical past and alter logs
- Wonderful-grained entry management and audit trails
- Integration of lineage from exterior techniques
- Implementation of stringent permission frameworks to forestall unauthorized information entry
Past information processing, CDRs are essential for sustaining information of information validation processes. Validation checks ought to be version-controlled in a code repository, permitting a number of variations to coexist and hyperlink to completely different research. Databricks helps Git repositories and established CI/CD practices, enabling the implementation of a strong validation examine library.
This method to CDR implementation on Databricks ensures information integrity and compliance and gives the flexibleness and scalability wanted for contemporary medical information administration.
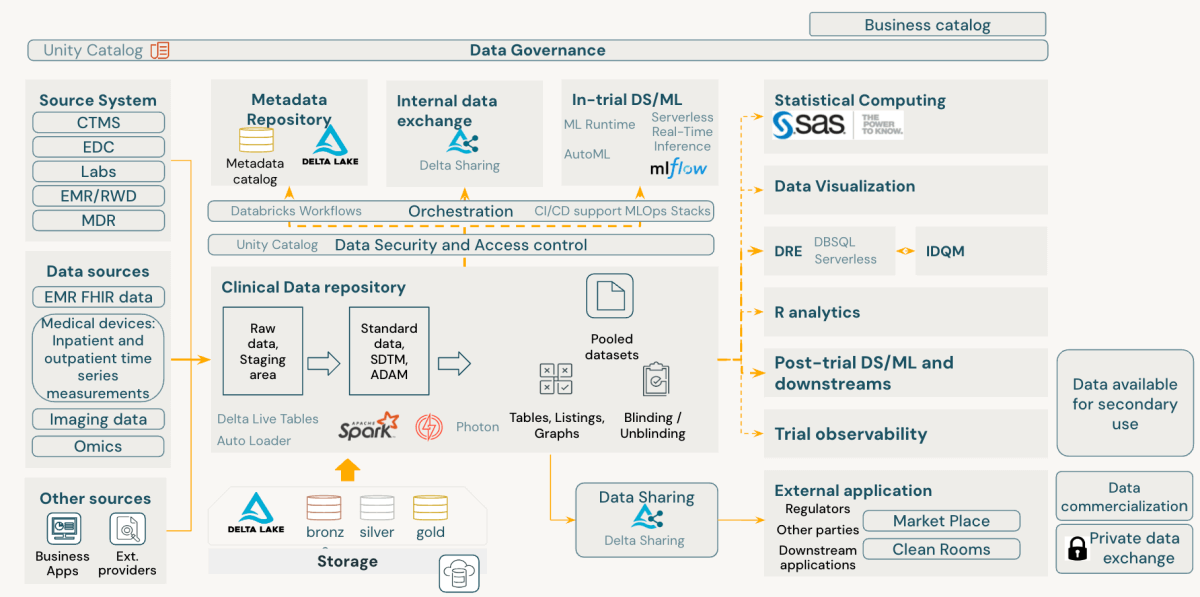
The Databricks Knowledge Intelligence Platform inherently aligns with FAIR ideas of scientific information administration, providing a sophisticated method to medical improvement information administration. It enhances information findability, accessibility, interoperability, and reusability whereas sustaining strong safety and compliance at its core.
Challenges in Implementing Trendy CDRs
No new method comes with out challenges. Medical information administration depends closely on SAS, whereas modem information platforms primarily make the most of Python, R, and SQL. This clearly introduces not solely technical disconnect but in addition extra sensible integration challenges. R is a bridge between two worlds — Databricks companions with Posit to ship first-class R expertise for R customers. On the identical time, integrating Databricks with SAS is feasible to assist migrations and transition. Databricks Assistant permits customers who’re much less acquainted with the actual language to get the assist required to put in writing high-quality code and perceive the prevailing code samples.
An information processing platform constructed on prime of a common platform will at all times be behind in implementing domain-specific options. Robust collaboration with implementation companions helps mitigate this threat. Moreover, adopting a consumption-based value mannequin requires additional consideration to prices, which should be addressed to make sure the platform’s monitoring and observability, correct consumer coaching, and adherence to finest practices.
The most important problem is the general success charge of a lot of these implementations. Pharma firms are continuously trying into modernizing their medical trial information platforms. It’s an interesting space to work on to shorten the medical trial length or discontinue trials that aren’t more likely to develop into profitable quicker. The quantity of information collected now by the typical pharma firm accommodates an unlimited quantity of insights which are solely ready to be mentioned. On the identical time, the vast majority of such tasks fail. Though there isn’t a silver bullet recipe to make sure a 100% success charge, adopting a common platform like Databricks permits implementing CDR as a skinny layer on prime of the prevailing platform, eradicating the ache of frequent information and infrastructure points.
What’s subsequent?
Each CDR implementation begins with the stock of the necessities. Though the {industry} follows strict requirements for each information fashions and information processing, understanding the boundaries of CDR in each group is important to make sure venture success. Databricks Knowledge Intelligence Platform can open many further capabilities to CDR; that’s why understanding the way it works and what it presents is required. Begin with exploring Databricks Knowledge Intelligence Platform. Unified governance with Unity Catalog, information ingestion pipelines with Lakeflow, information intelligence suite with AI/BI and AI capabilities with Mosaic AI shouldn’t be unknown phrases to implement a profitable and future-proof CDR. Moreover, integration with Posit and superior information observability functionally ought to open up the potential for taking a look at CDR as a core of the Medical information ecosystem reasonably than simply one other a part of the general medical information processing pipeline.
Increasingly more firms are already modernizing their medical information platforms by using trendy architectures like Lakehouse. However the massive change is but to return. The enlargement of Generative AI and different AI applied sciences is already revolutionizing different industries, whereas the pharma {industry} is lagging behind due to regulatory restrictions, excessive threat, and value for the flawed outcomes. Platforms like Databricks enable cross-industry innovation and data-driven improvement to medical trials and create a brand new mind-set about medical trials usually.
Get began right this moment with Databricks.
Quotation:
[1] Medical Trials Statistics 2024 By Phases, Definition, and Interventions
[2] Lu, Z., & Su, J. (2010). Medical information administration: Present standing, challenges, and future instructions from {industry} views. Open Entry Journal of Medical Trials, 2, 93–105. https://doi.org/10.2147/OAJCT.S8172
Study extra concerning the Databricks Knowledge Intelligence Platform for Healthcare and Life Sciences.

